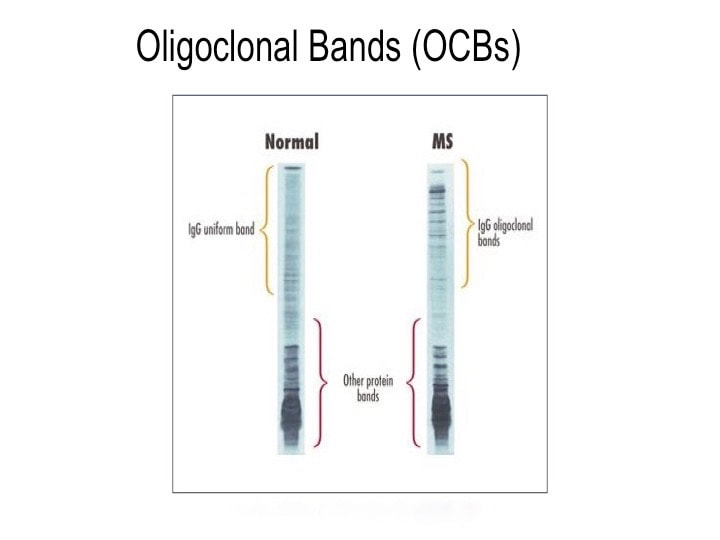
It includes proteins, electrolytes, antibodies, hormones, and sometimes outside agents like drugs and microorganisms. Doctors sometimes examine the antibodies present in patients’ serum to search for invading germs. And serology is a lousy way to diagnose disease. It’s inexact and not always timely. Antibodies are those little Y-shaped proteins made by the immune system (B-lymphocytes to be exact) that stick like glue to foreign invaders like viruses, bacteria, and other microbes. But they keep their cards close to their chests. For instance, Lyme disease takes weeks or even months to make antibodies against Borrelia burgdorferei, the bacterium that causes it. And infections with other bacteria can cause false positive tests for Lyme disease—even if those other infections occurred in the distant past. Call it “diagnostic archaeology,” because who really cares if you had Rocky Mountain spotted fever or some other infection years ago? You either died from it, or resolved it and live on. In multiple herpes cases, I have helped patients who were infected with herpes simplex virus (because I cultured it), yet their blood never made antibodies against the virus. Are those patients still infected with herpes or not? Actually, it’s not clear. You can see how how serologic diagnosis can lead to problems: over-diagnosis (false positives, as in Lyme testing) and under-diagnosis (false negatives, as in herpes testing). Sometimes serology tests are the best we can do. The syphilis agent, Treponema pallidum, has never been cultured in the lab. So if you want to figure out whether a patient has syphilis or not, you have to rely on serologies. And sometimes you just have to guess because patients with primary (the earliest form of) syphilis often don’t have a positive serologic because their immune system hasn’t had time to develop antibodies against the syphilis bacterium. Antibodies come in several forms. The most important is immunoglobulin G (IgG), which accounts for most antibodies in the blood and tissues. Other antibody types include IgA, which present in the gut and other mucosal surfaces, and IgE, responsible for many allergic responses. IgG immune responses to an infection are usually polyclonal, derived from many B-lymphocyte clones, each making slightly different antibodies, but all directed at one target. In contrast, B-lymphocyte cancers (myelomas) make only a single antibody species—a monoclonal. Oligoclonal (OCB) responses are neither monoclonal or polyclonal, but something in between with a few (oligo) B-lymphocytes producing a limited number of antibodies. OCBs are everywhere in MS patients’ spinal fluid and we have no idea why. Yet. Spinal fluid in healthy people contains very few antibodies—typically about 1/1000th the level in the blood. Past MS researchers tried to detect antibodies to a number of microbes, both in blood and spinal fluid. Wally Tourtellotte at UCLA led this effort, looking for antibodies in the spinal fluid of MS patients and comparing them to those found in the patients’ blood. To make a very long and complicated story short, his team basically found that MS patients are a little better at making antibodies compared to healthy control patients. But MS patients are not enough better at making antibodies to really account for their disease. And high levels of IgG antibody levels are often found in the spinal fluid of MS patients.
So what does spinal fluid serology have to do with MS? Everything, actually. Because “oligoclonal bands” are a terrible, confusing, and wordy way to describe IgG antibodies that collect in the spinal fluid. That’s right, most MS patients make IgG antibodies in their brain or spinal cord, and these collect in the spinal fluid. We can detect the presence of oligoclonal bands by a specialized test that uses an isoelectric focusing gel. Since all IgGs are about the same size, we use an electric charge to separate and visualize different antibody clones (also called species). These oligoclonal bands in the spinal fluid of MS patient are a hallmark of the disease – 90% of MS patients have them. When I was a medical student at Washington University in St. Louis in the 1980s, neurologist Charlie Trotter made big waves. He helped show that OCBs are commonly found in the spinal fluid of MS patients. At first, everyone believed these OCBs would be directed against myelin or some other human protein found in the brain, confirming the autoimmune nature of MS. Wrong! Sorry folks, these OCBs are not directed against myelin, myelin oligodendrocyte glycoprotein (MOG), or any other neural protein. In fact, 30+ years later no one really knows what these ever-present oligoclonal bands are doing. Oh sure, you can turn up the gain and show that some bands (antibody species) bind to Epstein Barr virus, or human herpes virus 6, but don’t believe that that’s the whole OCB enchilada. The actual targets of OCBs are still largely unknown. My lab team at the University of Utah intends to find out. Many MS researchers now believe that these OCBs are “poorly directed.” That is, they have no specific target but are produced haphazardly by the autoimmune process. I say these researchers are jumping to hasty conclusions. Because OCBs can also be found in the spinal fluid of patients with other diseases, particularly those involving neurologic infections. That is, patients with trypanosomiasis (African sleeping sickness), herpes simplex encephalitis, HIV, neurologic Lyme disease, neurologic syphilis, and neuroinvasive West Nile virus infection all have OCBs in their spinal fluid. It is logical that the targets of these OCBs are infecting or triggering organisms. It is predicted that the oligoclonal bands in specific MS patients are directed against microbes found in the brain, which our research has shown are often anaerobic bacteria (see the Microbes in MS sequencing paper recently published in Scientific Reports). MS patients need doctors and researchers to answer the biggest question: What are these tangled OCBs attacking in MS patients? Why are they there? What is their calling? Because once we know what the oligoclonal bands are aiming at, we can begin to intercede effectively. Said another way, the targets of these oligoclonal bands are the key to discovering the cause of MS. Royalty-free stock illustration ID: 81342454
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorDr. John Kriesel is Associate Professor of Infectious Diseases at the University of Utah School of Medicine. He began this blog to raise awareness and generate discussion about the possible causes of multiple sclerosis. Archives
March 2021
Categories |
 RSS Feed
RSS Feed